Nymox announces major results in long-term prostate treatment studies
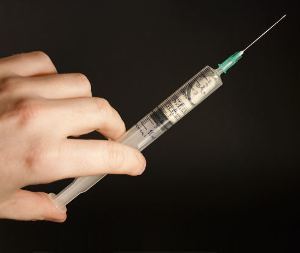
The seven years of follow up on the Phase III trials showed that BPH patients treated with fexapotide had only a 1.3 percent incidence of later prostate cancer development. In contrast, patients treated with PDE5 inhibitor drugs had a rate of 19.5 percent and the control subjects had a rate of 22.7 percent of cancer developing after four years.
Nymox also conducted a study using fexapotide as a treatment for low grade localized prostate cancer. Of the 147 participants, the patients treated with fexapotide showed better outcomes than the control subjects after 18 months.
Nymox will file for approval of fexapotide within the next two quarters. The low cost of producing the medication and success of the two Phase III trials puts Nymox ahead of its competition in the market for safe and effective prostate cancer injectable medications.
"Our extremely low cash requirements and strong, comprehensive successful clinical trial results add up to a very strong position for the company's future," Nymox CEO Dr. Paul Averback said. "In this context we furthermore look forward to reporting in the very near future on some additional new clinical trial results for fexapotide."







 Alerts Sign-up
Alerts Sign-up