ASHP and CMS partner to provide online hospital surveyor training course
All 2,000 CMS state surveyors must take the three-part training course. Read More »
ASHP announces launch of Standardize 4 Safety program
Initiative aims to improve IV and oral liquid medication safety. Read More »
CMS releases Medicare Advantage data sorted by racial and ethnic groups
Quality of care data shows disparities in care for African-American and Hispanic patients. Read More »
ASHP partners with ABIM in the 'Choosing Wisely' campaign
Initiative aims to help patients and health care providers avoid waste and risk of unnecessary treatments. Read More »
FDA approves drug to treat Parkinson's disease hallucinations and delusions
Nuplazid has been approved to treat the hallucinations and delusions experienced by some Parkinson's disease patients. Read More »
First generic version of Crestor approved
The FDA approved the first generic version of Crestor last week. Read More »
Proposed amendment to drug plan bill could spike costs for Oklahoma consumers, says national healthcare expert
Oklahoma consumers enrolled in health plans with insurance deductibles that are yet to be reached and those who are uninsured may bear the high costs of rising drug prices, thanks to a proposed piece of legislation that would give pharmacies permission to disregard negotiated discounts with insurers and transfer the price hikes on to consumers. Read More »
ASHP launches certificate programs for pharmacists
For its online continuing education program, "The Teaching Certificate for Pharmacists" is the first offering. Read More »
ASHP announces 2016 Fellows
Thirty-seven pharmacists were honored for excellence in pharmacy practice. Read More »
CMS launches Comprehensive Primary Care Plus model
CPC+ supports primary care doctors; promotes high-quality care for patients. Read More »
Surgeon General kicks off national tour in Baltimore
Dr. Vivek Murthy started his prescription drug crisis tour in Baltimore on April 5. Read More »
2016 ASHP Match program finds success
The American Society of Health-System Pharmacists (ASHP) second Match program concluded this week, with 4,273 individuals matched with more than 2,000 pharmacy residency programs nationwide. Read More »
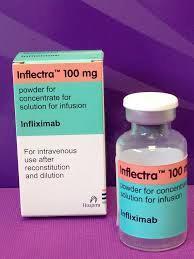
FDA approves Inflectra for IV infusion
Victims of digestive disorders, arthritis and psoriasis are one step closer to relief as of April 5, when the U.S. Food and Drug Administration approved Inflectra to address Crohn’s, colitis, and other chronic ailments. Read More »
ASHP announces 2016 ASHP Student Leadership Award recipients
The American Society of Health-System Pharmacists (ASHP) has acknowledged the accomplishments of 11 pharmacy students for their leadership on campus and in the pharmacy practice setting by awarding them with the ASHP Student Leadership Award. Read More »
ASHP’s annual midyear meeting wins multiple awards
The American Society of Health-System Pharmacists’ (ASHP) Midyear Clinical Meeting and Exhibition was recently honored with three awards. Read More »
FDA grants Actinium’s Iomab-B orphan drug designation
The U.S. Food and Drug Administration (FDA) has granted orphan drug designation for Iomab-B, a radioimmunotherapeutic developed by Actinium Pharmaceuticals that is designed to aid patients with reverted and stubborn Acute Myeloid Leukemia (AML) so they can receive a Hematopoietic Stem Cell Transplant (HSCT). Read More »
UsAgainstAlzheimer’s confident in new line of Alzheimer’s drugs
Recent research conducted by ResearchersAgainstAlzheimer’s (RA2), an UsAgainstAlzheimer’s (UsA2) network, reports 17 new drugs aimed at treating Alzheimer’s will be developed in the next five years. Read More »
FDA approves Teva’s CINQAIR Injection for asthma treatment
Teva Pharmaceutical Industries has announced that the U.S. Food and Drug Administration (FDA) has approved its interleukin 5 antagonist monoclonal antibody (lgG4 kappa), CINQAIR (reslizumab) Injection. Read More »
Allergan files ANDA for generic version of Abraxane
Allergan has announced that it has submitted an Abbreviated New Drug Application (ANDA) with the U.S. Food and Drug Administration (FDA) for the approval of market Paclitaxel Protein-Bound Particles for Injectable Suspension, 100 mg/vial. Read More »


















 Alerts Sign-up
Alerts Sign-up